Past Issues
Impact of the Addition of Sugar Beet Lime Sludge on the Composting of Argan Oil By-Products
Meriem Mobaligh1, Omar Saadani Hassani1, Youssef Ait Rahou2, and Khalid Fares1*
1Laboratory of Pharmacology, Neurobiology, Anthropobiology and Environment, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakech, Morocco
2Laboratory of Agro-Foods, Biotechnologies and Valorisation of Bioressources Vegetales, Department of Biology, Faculty of Sciences Semlalia, Cadi Ayyad University, Marrakesh, Morocco
*Corresponding author: Khalid FARES, Laboratory of Pharmacology, Neurobiology, Anthropobiology and Environment, Faculty of Sciences Semlalia, Cadi Ayyad University, P.O. Box: 2390, 40001 Marrakech, Morocco; Email: [email protected]
Received Date: October 20, 2023
Publication Date: October 27, 2023
Citation: Mobaligh M, et al. (2023). Impact of the Addition of Sugar Beet Lime Sludge on the Composting of Argan Oil By-Products. Catalysis Research. 3(3):17.
Copyright: Mobaligh M, et al. © (2023).
ABSTRACT
The argan tree is a multi-purpose tree. Every part of the tree is a source of income or food for the user. The extraction of argan oil from the fruit results in three by-products: the shell, the pulp and the cake. The pulp and meal are mainly used as fodder or as a source of heat, while the shell is the most competitive renewable energy source for the production of heat and electricity. In this study, and for the first time in this field, we set out to test the recovery of these by-products, in particular pulp and cake, through composting while using sugar lime sludge.
The physicochemical monitoring during the composting process revealed that the temperature of the three windrows followed a typical evolution, including a long thermophile phase that was maintained for more than two months at the level of the three windrows. The humidity was maintained at the ideal composting level (40 %) through regular watering, to ensure good microbial activity. The degradation of organic matter is better in the presence of a beet sugar lime sludge 30 % (33.94 %) compared to (16.15 %) and (5.95 %) for windrows based on 15 % beet sugar lime sludge and the control, respectively. The pH of windrows based on this agri food waste was corrected from the start of composting compared to the control windrow due to the alkaline character of the beet sugar lime sludge. In terms of final quality, the composts produced from the beet sugar lime sludge comply with the NFU 44-051 standard. A germination index higher than 50% showed the absence of any phytoxicity. As well as for the humification parameters suggest that these two composts are mature and well humified compared to the control compost.
Our results provide a sustainable solution for the recovery of argan by-products and carbonation lime from the sugar industry, thus promoting environmental protection and the circular economy.
Keywords: beet sugar lime sludge; composting; physicochemical parameters; organic matter; phytotoxicity; humification parameters
INTRODUCTION
Argan oil has always been a luxury product and a symbolic production of Morocco. Currently, its reputation has spread widely and, in recent years, this oil is sold and appreciated in several countries, because of its organoleptic characteristics and nutritional properties.
However, the companies producing argan oil and cosmetic products made from argan also generate a huge quantity of waste and by products resulting from the extraction of argan oil from the fruit such as pulp and oil cake. Similarly, to the classic operations of production and marketing, this industry must be strengthened by research work in order to enhance and to contribute to the success of the transformation of a traditional production system, to a modern production system, taking into account the local constraints and the protection of the environment.
In this study, and for the first time, we were interested in valorizing these by-products, especially the pulp and oil cake, through composting which is considered a powerful tool to conserve and recycle organic matter, and it also allows obtaining stabilized products with high agronomic value (Tognetti et al., 2011) [1]. To do this, we optimized the windrow composting of argan fruit by-products (pulp and oil cake) which is characterized by an acidic pH with the sugar lime sludge. The addition of the sugar lime sludge in our study is based on their basic pH which gives them the role of an alkaline agent and their richness in calcium (Asadi, 2006) [2].
The valorization of its by-products will be done by a simple process and accessible to the Moroccan population which exploits the argan tree. The substrate introduced for the preparation of composts and produced in excess by the sugar industry, it represents almost 3% of the quantity of sugar cane and 8% of the treated sugar beets (Etahiri et al., 2006) [3], in addition does not present any risk on the environment and the health of the population. It should be noted that this huge quantity of sugar lime sludge is increasing year by year with the increase of sugar beet and sugar cane production in Morocco. These sugar lime sludges are rejected outside the factories in wild lands without any valorization or treatment. In fact, Moroccan sugar factories set aside a budget for each campaign devoted solely to the evacuation of the sugar lime sludge that accumulates every year on the outskirts of the factories. The search for solutions that could allow the reuse of by-products or waste as secondary raw materials has become a major concern for the different sectors of the industry (Vaccari et al., 2005) [4].
The advantage of this present work, it allows us to engage in the challenge of the transition to a circular economy model in order to face the depletion of the planet's resources. Thus to leave the linear model "make, consume, throw away" and move towards a circular economy.
MATERIALS AND METHODS
Selection and characterization of waste
The determination of the waste composition is an essential step for a successful composting. Three substrates were used to prepare three different windrows: pulp (P), oil cake (OC) and sugar lime sludge (SLS). The main physicochemical characteristics of the raw materials used to prepare the composts are presented in Table 1.
Table 1: Physicochemical characteristics and origin of the different wastes used in our study.
|
Characteristics |
SLS |
P |
OC |
|
Humidity (%) |
4,7±0.05 |
9.74±0.06 |
6.08±0.04 |
|
OM (% DM) |
12.73±0.15 |
98.51±1.03 |
95.85±0.32 |
|
pH |
8,07±0.08 |
5.41±0.01 |
4.02±0.02 |
|
Nitrogen (g/kg de DM) |
0.24±0.06 |
2.4±0.05 |
0.4±0.01 |
|
Origin |
Doukkala Sugar Factory |
Cosmetics company |
|
|
Nature |
Lime sludge from sugar beet juice purification |
Argan fruit pulp |
Residue from argan oil extraction |
The pulp and the oil cake have a high percentage of organic matter in the order of 95.85% DM for pulp and 98.51% DM for oil cake; on this basis, we can qualify these two substrates as good materials for composting. But they showed an acidic pH incompatible with good bacterial activity during composting, that's why we have incorporated the sugar lime sludge which are characterized by a basic pH. Concerning heavy metals, which are very toxic pollutants and inhibitors of plant germination, the sugar lime sludge are free of them (Table 2). The concentrations of heavy metals are low and lower than the normative limits recommended in (NF U44-051) [5].
Table 2: Mineralogical characteristics of the sugar lime sludge used in this study.
|
Elements |
Concentration (mg/kg of DM) |
|
Ag |
Trace |
|
Al |
6770 |
|
As |
trace |
|
B |
30 |
|
Ba |
20 |
|
Ca |
82640 |
|
Cd |
Trace |
|
Co |
Trace |
|
Cr |
20 |
|
Cu |
30 |
|
Fe |
5150 |
|
K |
4480 |
|
Mg |
19500 |
|
Mn |
290 |
|
Na |
11120 |
|
Ni |
Trace |
|
P |
9400 |
|
Pb |
Trace |
|
Si |
870 |
|
Sn |
200 |
|
Ti |
340 |
|
Zn |
80 |
Preparation of Composts
Proportions of the windrows
The composting trail was corresponding to three windrows W1, W2 and W3 which were monitored for 110 days. The proportions of different mixtures of the initial substrates (Kg) used are presented in Table 3. The percentage of sugar lime sludge was 15 % and 30 % in W2 and W3, respectively. Each windrow is placed on impermeable plastic sheets (polypropylene) to avoid runoff and leaching phenomena during the wetting of the mixtures.
Table 3: Composition of the different windrows (Kg).
|
Windrows |
W1 |
W2 |
W3 |
|
SLS |
0 |
16 |
36 |
|
P |
70 |
65 |
60 |
|
OC |
28 |
26 |
24 |
Monitoring of the Composting
Manual turning is done every two days for the first two weeks, then once a week for the rest of the process to ensure the aeration of the windrows which conditions the activity of the microorganisms. The humidity of the windrows is also controlled by watering and covering or uncovering the windrow in order to have optimal values between 50 and 60% throughout the composting process. In addition, a uniform sampling is carried out on the whole windrow, on the surface and at different depths, in order to obtain a homogeneous and representative sample to be used for physicochemical analyses.
Physicochemical Analyses
The samples taken from the three windrows were characterized according to the main conventional physicochemical analyses followed during the composting process.
Temperature measurement was performed daily for the first two weeks and then once or twice a week until the end of the composting process. Moisture is determined by the drying method in an oven at 105 ± 2°C for 24 h until a constant weight is obtained. The organic matter content (OM) was determined after calcination in an oven at 650°C for 6 hours. The total organic carbon content (TOC) was calculated by the equation of Haug (1993) [6].
TOC (%) = OM (%) / 1.8 (1)
The pH of the different samples were measured in a 1:10 (w/v) aqueous extract. Total Kjeldahl nitrogen was determined according to the Kjeldahl method. All analyses were performed in three replicates.
The main parameters related to phytotoxicity of composts were also characterized such as germination percentage (%G), Germination Index (GI) according to the method of Zucconi et al. (1981) [7].
Calculating the humification indices (humification index (HI), humification rate (HR), and degree of humification (HD), which are based on the quantification of the humic fraction relative to the fulvic fraction, was used to verify the maturity and stability of the composts produced. Humic compounds were isolated from the three composts using Schnitzer's method (1982) [8], following which we separated humic (HA) and fulvic (FA) acids by acidification with a sulfuric acid solution (6 N). Equations 2 (Ciavatta et al., 1988) [9], 3 (Sequi et al., 1986) [10], and 4 (Sugahara and Inoko, 1981) [11] are used to determine the humification rate, humification degree, and humification index, respectively:
HR% = (CTHM / TOC) × 100 (2)
HD% = (CTHM / CH) × 100 (3)
HI = HAC / FAC (4)
Where,
CH: Carbon of humus; CTHM: Carbon of total humic matter; HAC: Humic acid carbon; FAC: Fulvic acid carbon.
Statistical Analysis
All the data are expressed as the mean and standard deviation. The results were analyzed by the ANOVA using XLStat Premium, version 2013.
RESULTS AND DISCUSSION
Temperature
In the three windrows, the temperature evolution (Figure 1) reveals a decent evolution of the composting process. From the first day after the commencement of composting, all windrows entered the thermophilic phase, indicating rapid degradation of easily degradable organic matter, which served as a primary source of nutrients for the microorganisms (Wang et al., 2013) [12]. This long-term thermophilic phase could be attributed to the high organic matter content in the initial substrates employed, as well as the existence of complex molecules that progressively disintegrate into simpler molecules with the creation of heat (Awasthi et al., 2016) [13]. Both windrows 1 and 3 gradually reverted to the mesophilic phase after 77 days, while windrow 2 stayed high for 28 days longer than the other two windrows. The temperature of the windrows was reduced to ambient temperature after this phase, demonstrating that the humification phenomena is in control and the composts produced are mature
Figure 1: Temperature variation during the composting.
Humidity
Figure 2 illustrated the three windrows' moisture variations throughout the composting process. The ANOVA revealed that there was no significant variation in moisture evolution across the windrows during the composting process based on the concentration of the sugar beet lime sludge. Because the original substrates were too dry, the moisture content was very low at the start of composting (40.1 % for W1, 40.3 % for W2, and 40.4 % for W3), but it increased to the optimum moisture content needed during the process due to the addition of water.
Figure 2: Variation of the humidity in the windrows during composting process.
pH
pH is one of the most important factors that influence microbial activity throughout the composting process (Awasthi et al., 2016) [13]. According to Lopez-Cano et al. (2016) [14], pH oscillations during the composting process are caused by changes in the chemical composition of the initial substrates. The addition of sugar beet lime sludge to initial substrate had a substantial impact on the pH of the windrows 2 and 3 during the composting process. As a result, the pH of the windrows treated with sugar beet lime sludge was significantly corrected as soon as the composting process started. The ANOVA revealed that the addition and concentration of sugar beet lime sludge had highly significant impacts. The high pH value for windrow 3 with sugar beet lime sludge concentration (30%) was owing to the large amount of sugar beet lime sludge, which had a remarkable effect on the composting mixture's pH. W1 had a pH of 7.70, W2 had a pH of 8.45, and W3 had a pH of 8.72. The current findings are also in line with the findings of Saadani et al. (2019) [15], who found that the sugar beet lime sludge based windrows had a slightly higher pH than the control windrow.
Figure 3: Evolution of the pH in the different windrows during the composting.
ORGANIC MATTER
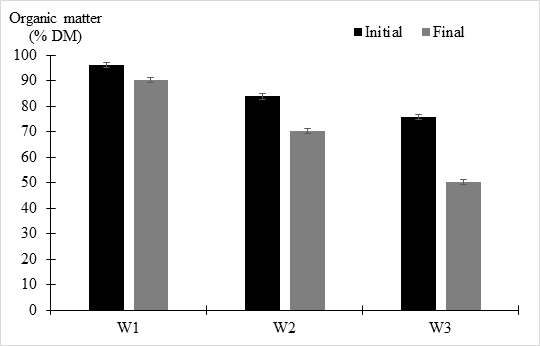
Figure 4 depicted the rate of organic matter decomposition during the composting process. The degree of organic matter degradation was a clear indicator of the composting process success (Wang et al., 2016) [16]. Due to the addition of the sugar beet lime sludge and their varying concentrations, the initial proportion of organic matter was gradually modified during the composting process. The addition of sugar beet lime sludge and its concentration degradation process is reflected in the decrease in organic matter (Waqas et al., 2018) [17]. The results revealed that maximum organic matter decomposition occurred during the thermophilic phase, which could be linked to high microbial activity (Sun et al., 2016) [18]. The control windrow has a higher percentage of organic matter than the other windrows, indicating less organic matter degradation and a considerable amount of inaccessible organic matter. The reduced rate of organic matter decomposition in windrow 1 (5.96 %) corresponds to a lower pH, resulting in anaerobic conditions and a poor composting process. Normal microbial activities were inhibited by the acidic pH of the control windrow, which also the control windrow's acidic pH limited normal microbial activities, as well as the decomposition of organic acids produced (Waqas et al., 2018) [17]. Furthermore, there was no alkaline amendment to buffer the acidic environment, resulting in a drop in pH and an unfavorable setting for microbial activity (Waqas et al., 2018) [17]. The final compost's organic matter content was as follows: W1 (90.39 %), W2 (70.37 %), W3 (50.23 %). However, it was discovered that a higher concentration (30%) of sugar beet lime sludge resulted in faster degradation of organic waste. had a significant influence on the degradation of organic matter when compared to the control, according to ANOVA and Tukey's HSD test for multiple comparisons. W3 had the greatest reduction in organic matter (33.94 %), followed by W2 (16.15 %). The efficiency of the
Figure 4: Organic matter contents in the different windrows.
C/N
Compost maturity is determined by the C/N ratio (Colón et al., 2011) [18]. The C/N ratios in the windrows 2 and 3 decline quicker than in the control windrow (Figure 5). The addition of sugar beet lime sludge an alkaline ingredient may have aided biological activity growth. Windrows 1 and 2 had the highest C/N ratio 21.19 and 21.48 respectively after 110 days of biodegradation, followed by windrow 3 (16.71). A C/N ratio of less than 20 has been cited by many authors as one of the indicators of compost maturity (Nolan et al., 2011) [19]. According to this criterion, windrow W3 reached maturity at 110 days, whereas W1 and W2 did not reach maturity at all. Due to the large availability of easily biodegradable compounds for bacteria, the loss of organic matter increases with the time of biodegradation (Benito et al., 2003) [20].
Figure 5: Evolution of the C/N ratio during composting process.
Final Quality of the Composts
Humification Process during Composting
Compost quality is proven by the quantity of stable humus formed after the biodegradation of organic matter (Domeizel et al., 2004) [21], hence monitoring humification parameters during composting could be a typical measure of the evolution of compost maturity and stability, according to (López et al., 2002) [22]. Table 4 shows that at the end of the composting process (110 days), all composts including sugar beet lime sludge had a degree of humification and a humification rate higher than 70% and 35%, respectively, which are considered norms, indicating good humification (Ciavatta et al., 2001) [23]. Also, composts 2 and 3 had a humification indices higher than the 1.6 value for stabilized organic matter estimated by (Jimenez and Garcia, 1992) [24]. The results of the control, suggesting that organic matter degradation was less in the control and that the humic compounds produced were unstable.
Table 4: Humification indices of the final composts.
|
Composts |
C1 |
C2 |
C3 |
|
IH (CAF/CAH) |
1,43±0,06 |
0,5±0,05 |
0,28±0,05 |
|
IH (CAH/CAF) |
0,7±0,03 |
2,01±0,19 |
3,58±0,7 |
|
RH |
33,2±0,79 |
51,28±0,50 |
77,32±2,5 |
|
DH |
34,5±1,52 |
70,93±0,48 |
82,75±0,77 |
Phytotoxicity Test
Phytotoxicity test is the only approach to detect the toxicity of immature composts introduced into the soil. In the present study germination of maize seeds in the presence of the three compost extracts (Figure 6) was detected from the first day for both dilutions. The Petri dishes containing the compost extracts (1/25) reached germination percentages of about 72.5 %, 82.5 % and 95 % for C1, C2, and C3, respectively. While the dilution (1/50) had a slightly higher germination rate: 90 %, 85 % and 97.5 % respectively, for C1, C2, and C3. Because their germination rates were above 80% (Zucconi et al., 1981; Erhart and Burian, 1997) [7,25], C2 and C3 can be regarded mature and non-toxic, but C1, which did not include sugar beet lime sludge, did not attain maturity even after 110 days.
The maize germination indexes (figure 7) of the C2 and C3 were higher than 50% for both dilutions (1/50 and 1/25), indicating that there was no toxic. Compost is considered non-toxic when its germination index surpasses 50%, according to Zucconi et al. (1981) [7].
Figure 6: Effect of composts extracts (a: 1/25 dilution; b: 1/50 dilution) on the evolution of germination percentage of maize.
Figure 7: Germination index of maize in the presence of the compost extracts (a: 1/25 dilution; b: 1/50 dilution).
CONCLUSION
The results of this study provided further evidence that composting is a viable option for the recovery of argan tree by-products. The initial physico-chemical characteristics of the pulp and cake revealed that they are characterized by an acidic pH (4-5), requiring the addition of waste with a basic pH essential for the process to run smoothly. The monitoring of physico-chemical parameters throughout the composting period and the results of the phytotoxicity test as well as humification parameters revealed the stability and maturity of composts containing sugar beet lime sludge (C2 and C3) compared with the control (C1).
REFERENCES
- Tognetti C, Mazzarino MJ, Laos F. (2011). Comprehensive quality assessment of municipal organic waste composts produced by different preparation methods. Waste Manage. 31(6):1146-1152.
- Asadi M. (2006). Beet-Sugar Handbook. John Wiley & Sons, Hoboken:800.
- Etahiri S, Amine J, Bihaoui B. (2006). Production des déchets organiques biodégradables par l’industrie agroalimentaire au Maroc. MOROCOMP (LIFE TCY05/MA000141):12.
- Vaccari G, Tamburini E, Sgualdino G, Urbaniec K, Klemeš J. (2005). Overview of the environmental problems in beet sugar processing: possible solutions. J Cleaner Production, 13(5):499-507.
- NFU 44-051. Amendements Organiques—Dénomination, Spécifications et Marquage. 2006. Available online: https://www.boutique.afnor.org/fr-fr/norme/nf-u44051/amendements-organiques-denominations-specifications-et marquage/fa125064/754.
- Haug RT. (1993). The practical handbook of compost engineering. Lewis publishers, Boca Raton.
- Zucconi F, Forte M, Monaco A, Bertoldi MD. (1981). Biological evaluation of compost maturity. BioCycle. 22(2):27–29.
- Ciavatta C, Antisari LV, Sequi P. (1988). A 1st approach to the characterization of the presence of humified materials in organic fertilizers. Agrochimica. 32:510-517.
- Schnitzer M. (1982). Organic matter characterization. In: Methods of Soil Analysis, Part 2. Chemical and Microbiological Properties, 2nd (Ed), Page AL, Miller RH, Keeney DR. (Eds.). Agronomy Monograph, N°9. Am Society Agro. Madison, Wisconsin USA:581-595.
- Sequi P, De Nobili M, Leita L, Cercignani G. (1986). A new index of humification. Agrochimica. 30:175–180
- Sugahara K, Inoko A. (1981). Composition analysis of humus and characterization of humic acid obtained from city refuse compost. Soil Sci Plant Nutri. 27:213–224.
- Wang K, Li W, Gong X, Li Y, Wu C, Ren N. (2013). Spectral study of dissolved organic matter in biosolid during the composting process using inorganic bulking agent: UV–vis, GPC, FTIR and EEM. Int Biodeter Biodegr. 85:617-623.
- Awasthi MK, Wang Q, Huang H, Ren X, Lahori AH, Mahar A, et al. (2016). Influence of zeolite and lime as additives on greenhouse gas emissions and maturity evolution during sewage sludge composting. Bioresource Technol. 216:172-181.
- López M, Elorrieta M, Vargas-Garcı́a M, Suárez-Estrella F, Moreno J. (2002). The effect of aeration on the biotransformation of lignocellulosic wastes by white-rot fungi. Bioresource Technol. 81:123–129.
- Hassani OS, Saadaoui N, Loqman S, Rida S, Soraa N, R’zina Q, et al. (2019). Impact of carbonatation lime addition on composting of vegetable oil refining sludge. Sugar industry-zuckerindustrie. 144(3):139-146.
- Wang Q, Wang Z, Awasthi MK, Jiang Y, Li R, Ren X, et al. (2016). Evaluation of medical stone amendment for the reduction of nitrogen loss and bioavailability of heavy metals during pig manure composting. Bioresource Technol. 220:297-304.
- Waqas M, Nizami AS, Aburiazaiza AS, Barakat MA, Ismail IMI, Rashid MI. (2018). Optimization of food waste compost with the use of biochar. J Envir Manage. 216 :70-81.
- Sun D, Lan Y, Xu EG, Meng J, Chen W. (2016). Biochar as a novel niche for culturing microbial communities in composting. Waste Manage. 54:93-100.
- Nolan T, Troy SM, Healy MG, Kwapinski W, Leahy JJ, Lawlor PG. (2011). Characterization of compost produced from separated pig manure and a variety of bulking agents at low initial C/N ratios. Bioresource Technol. 102:7131–7138.
- Benito M, Masaguer A, Moliner A, Arrigo N, Palma RM. (2003). Chemical and microbiological parameters for the characterisation of the stability and maturity of pruning waste compost. Biology Fertility Soils. 37:184-189.
- Domeizel M, Khalil A, Prudent P. (2004). UV spectroscopy: a tool for monitoring humification and for proposing an index of the maturity of compost. Bioresource Technol. 94:177–184.
- López-Cano I, Roig A, Cayuela ML, Alburquerque JA, Sánchez-Monedero MA. (2016). Biochar improves N cycling during composting of olive mill wastes and sheep manure. Waste Manage. 49 :553-559.
- Ciavatta C, Francioso O, Montecchio D, Cavani L, Grigatti M. (2001). Use of organic wastes of agroindustrial and municipal origin for soil fertilisation: Quality criteria for organic matter. In: The First International Encounter, Management of Organic Wastes in Rural Mediterranean Areas. 22-23 February, University of Navarra, Pamplona, Spain:1-17
- Jimenez EI, García VP. (1992). Determination of maturity indices for city refuse composts. Agriculture, Ecosystems Envir. 38(4):331-343.
- Erhart E, Burian K. (1997). Evaluating quality and suppressiveness of Austrian biowaste composts. Compost Science & Utilization. 5(3):15-24.
 Abstract
Abstract  PDF
PDF